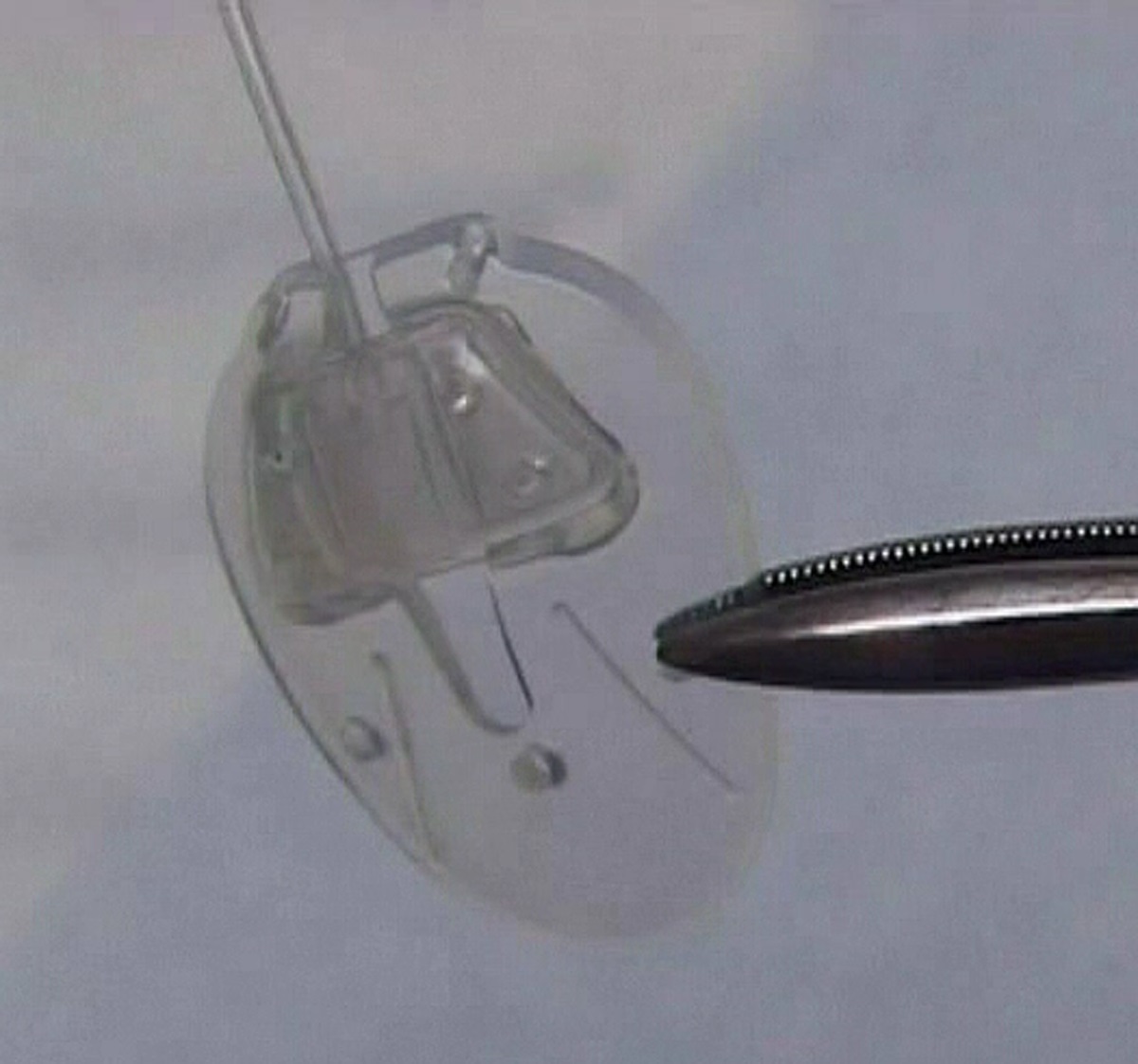
Aqueous shunt
FAQ
Frequently Asked Questions
Aqueous shunts are made up of a small silicone tube (less than 1 mm in diameter) attached to a plate. The tube takes the aqueous humor from inside the eye and drains it to the plate which sits on the white of the eye (sclera). The plate sits under the skin of the eye (conjunctiva), behind the eyelid. Aqueous shunts have various other names such as tube implants, glaucoma tube shunts, glaucoma drainage devices, glaucoma drainage implants and setons. These names all refer to the same thing. Although there are many types of shunt available, two brands are in common use today and they function in a similar fashion. These are called the Ahmed Glaucoma Valve and the Baerveldt Glaucoma Implant. In certain circumstances a third type, known as the Molteno Implant, might also be used.
Aqueous shunts are devices that are used to reduce the eye pressure (intraocular pressure) in glaucoma by draining the fluid (aqueous humor) from inside the eye to a small blister or bleb behind the eyelid. Reducing the pressure on the optic nerve in this manner prevents further damage and further loss of vision in glaucoma.
The surgery helps to reduce the build up of pressure in the eye and prevent further damage and loss of vision in glaucoma. It will not however restore vision already lost due to damage from glaucoma.
Most glaucoma surgical studies examine success rates over a 5 year period. With aqueous shunts such as the Baerveldt, the expected success rate over 5 years is now between 70 and 80%. Although a sizeable proportion of patients achieve good pressure control without the need for continued glaucoma medication, many patients still require some medication to assist the shunt in controlling the pressure. In such circumstances, the medication required is usually less than that required before the surgery.
The aqueous shunt itself is not normally visible on the outside of the eye. When the shunt is functioning normally, the drained fluid accumulates in a blister or bleb in the conjunctiva. The plate and its bleb are positioned far back behind the eyelid so they cannot usually be seen. Occasionally, the shunt or bleb can be seen in extremes of gaze, when the eye is looking very far down and in. A patch made of donor eye tissue, either from sclera (eye wall) or cornea (transparent tissue that forms the front of the eye) is often used to keep the shunt in place. This is the only part of the operation that might be visible after surgery.
Prior to undergoing surgery, patients are asked to continue all drops and tablets in accordance with their normal treatment regimen up until the morning of the operation. Blood thinning medications such as Aspirin, Warfarin and Clopidogrel should also be continued. Patients who are taking Warfarin are advised to have their level (eg. INR) checked at least 2 weeks prior to surgery to ensure it is within the correct therapeutic range.
If patients opt to have the surgery performed under general anaesthesia, a preoperative assessment of their general health will be carried out prior to the surgery. Underlying medical conditions including cardiac disease, uncontrolled high blood pressure or diabetes will need to be addressed prior to scheduling of surgery.
Aqueous shunt surgery may last one to two hours.
Anaesthesia
Aqueous shunt implantation is often performed under general anaesthesia, although local anaesthesia is also possible under certain circumstances.
Mitomycin C
During the surgery, Mitomycin C may be applied to the surface of the eye for a brief period of time (up to 5 minutes). Mitomycin C is a drug that was originally used to treat cancer, but it is also used in glaucoma surgery to reduce scarring. Scarring prevents the shunt from functioning in the long term, as it prevents the aqueous humor from being absorbed back into the circulation. The Mitomycin C is then washed away from the eye with sterile water so that no residual drug remains.
Donor patch
A patch made from tissue either from an eye bank (cornea or sclera) or from a commercial source (pericardium) is used to prevent break down of the conjunctival surface tissue over the shunt. If donor tissue is not used, breakdown of the conjunctival surface of the eye over the implant can occur in 10‐14% of cases. When donor tissue is used the risk of breakdown is less than 3%. The donor tissues used in aqueous shunt surgery are not live transplants. They are simply used to reinforce the eye surface over the outside of the shunt.
Patients are usually discharged home from hospital either the same day as the surgery or the following day. It is preferable to examine the eye again one day after surgery.
The eye is normally padded after surgery and the eye pad is removed the following day. If the unoperated eye does not see well, then the operated eye will not be padded. Instead, a clear shield will be placed on the operated eye so that it is still possible to see after surgery.
Eyedrops will be prescribed to use regularly after surgery. These are commenced on the day after surgery, after the post‐operative examination. It is not usually necessary to use eyedrops the first night after the surgery.
Acetazolamide (Diamox) tablets should also be stopped the night of surgery unless advised otherwise.
It is important that any eyedrops for the unoperated eye are continued unless advised otherwise.
Patients are usually seen once a week for the first 4 weeks, and may be seen more frequently if the eye pressure is either too high or too low.
In some cases, the ligature or occluding suture may cause high pressure after surgery. A ligature can be cut using a laser, usually 2‐3 weeks after surgery at which point the pressure drops. This procedure is very quick, painless and is performed in out‐patients. The occluding suture inside the tube can usually be removed after 3 months and occasionally before. The occluding suture can sometimes be removed in clinic but more often requires a return to the operating theatre to have it removed as a short operation.
Sometimes the pressure may be too low after surgery and this can sometimes be dangerous. Although very low pressure is often painless, it may be associated with a dull aching feeling or a throbbing sensation within the operated eye. Low pressure, when it occurs, is usually detected during clinic appointments and is often remedied by stopping any pressure‐lowering eye drops and reducing steroid eye drops. Sometimes an injection of a jelly material (viscoelastic) is required to raise the pressure. Occasionally, a further operation is needed to reduce the drainage from the tube.
Severe complications are uncommon but are most likely to happen if the eye pressure drops very low or very quickly in the early postoperative period. A very low or an abrupt drop in eye pressure can result in a choroidal haemorrhage (severe bleeding at the back of the eye). This happens in less than 1% of aqueous shunts. If the pressure drops very low it may be elevated again either using an injection of a viscoelastic gel or a gas into the eye in the clinic, or by a return to the operating theatre to have the tube adjusted. These interventions are only performed when the pressure is very low in order to prevent complications such as a choroidal haemorrhage, rather than waiting until after they occur.
The risk of serious infection inside the eye from aqueous shunt surgery is rare (<1%). There is also a small long‐term risk that the tube implant will develop a blockage (requiring further surgery to unblock the tube); erode (the surface conjunctiva over the shunt breaks down, requiring a repair operation); or rub against the cornea requiring further surgery to either move the tube so it does not rub or, in extreme cases where significant corneal damage has occurred, a corneal transplant may be required.
Editors
Founding Editors
MIGS.org was developed by glaucoma surgeons with a broad experience in minimally invasive glaucoma surgery to provide patients with evidence-based and unbiased information.


Glaucoma Specialist
Dr Nathan Kerr